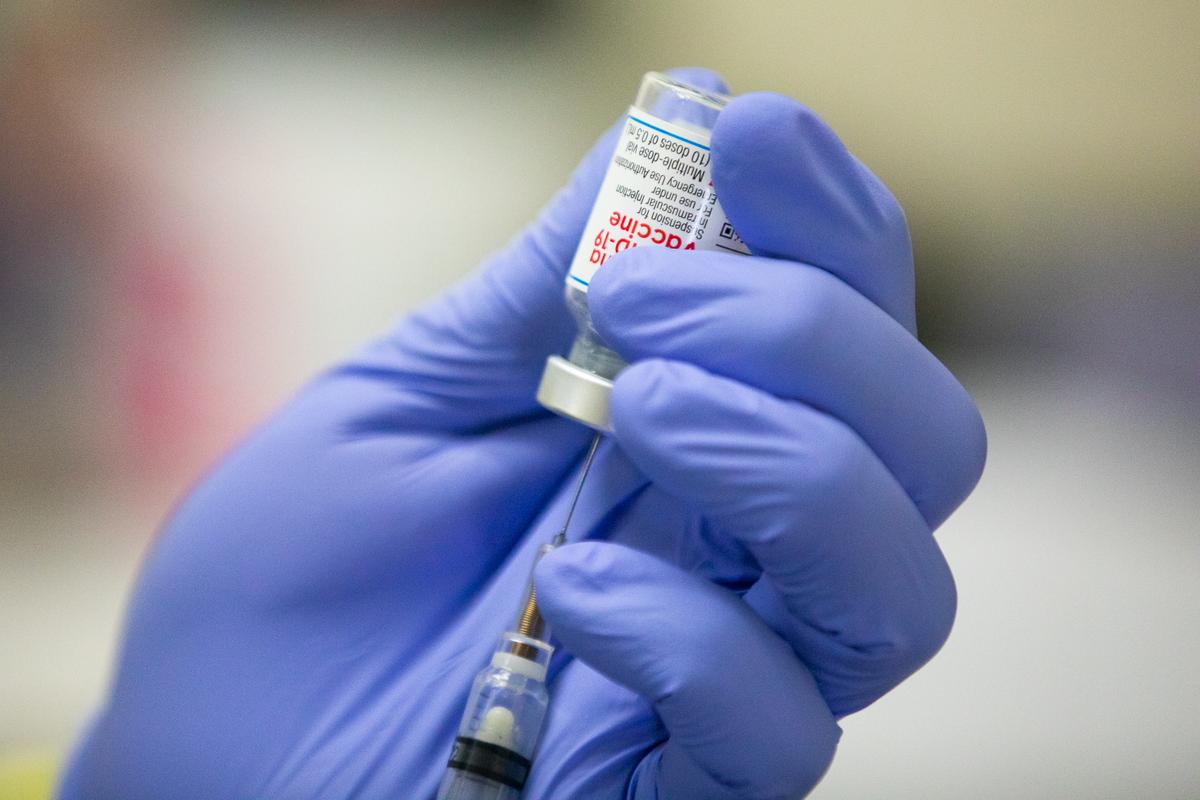
Regulators in the United States and elsewhere should not clear Moderna’s COVID-19 vaccine for children based on a clinical trial that found the shot under 50 percent effective in preventing infection, experts say.
Moderna is pushing for emergency authorization from the U.S. Food and Drug Administration (FDA) for its jab for kids as young as 6 months of age, based on the results of the trial.