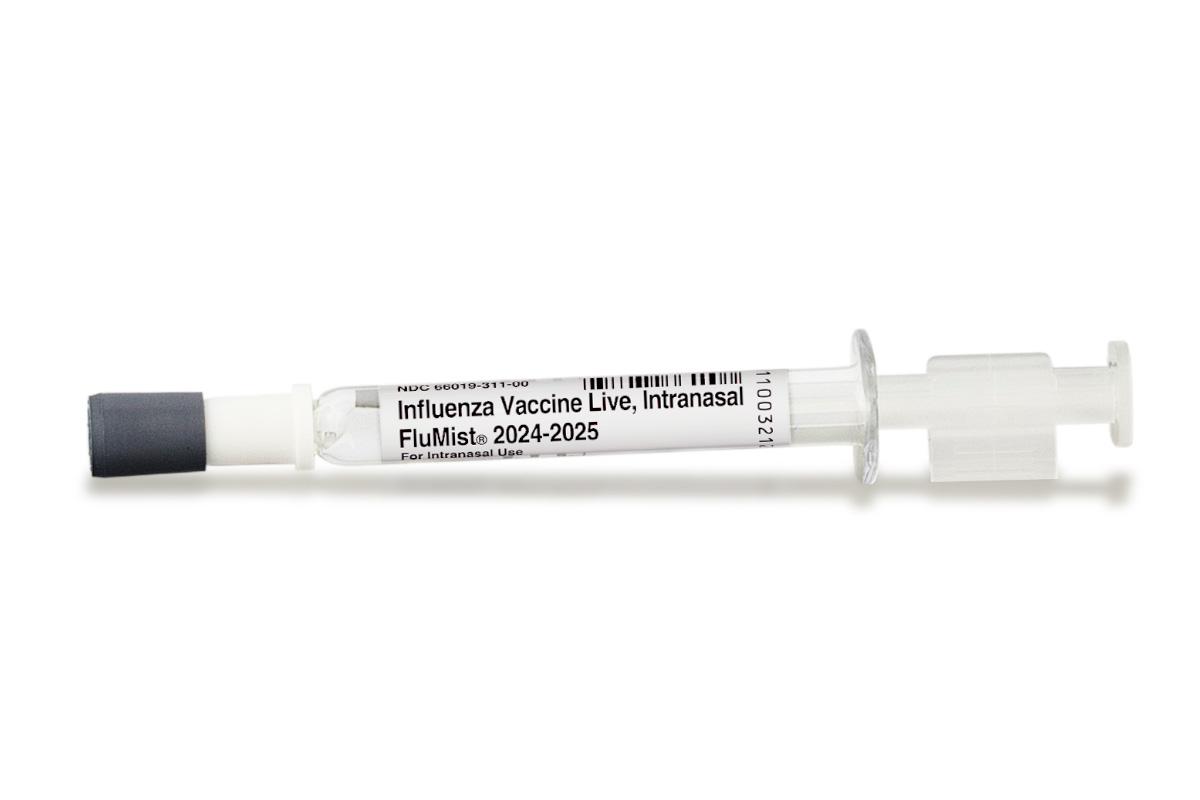
Experts share different views on the U.S. Food and Drug Administration’s (FDA) recent decision to expand its approval of FluMist, allowing the influenza nasal spray to be self-administered for the first time.
Pharmacists can prescribe the nasal vaccine and the spray can also be administered by a caretaker, the FDA said in a statement on Friday.