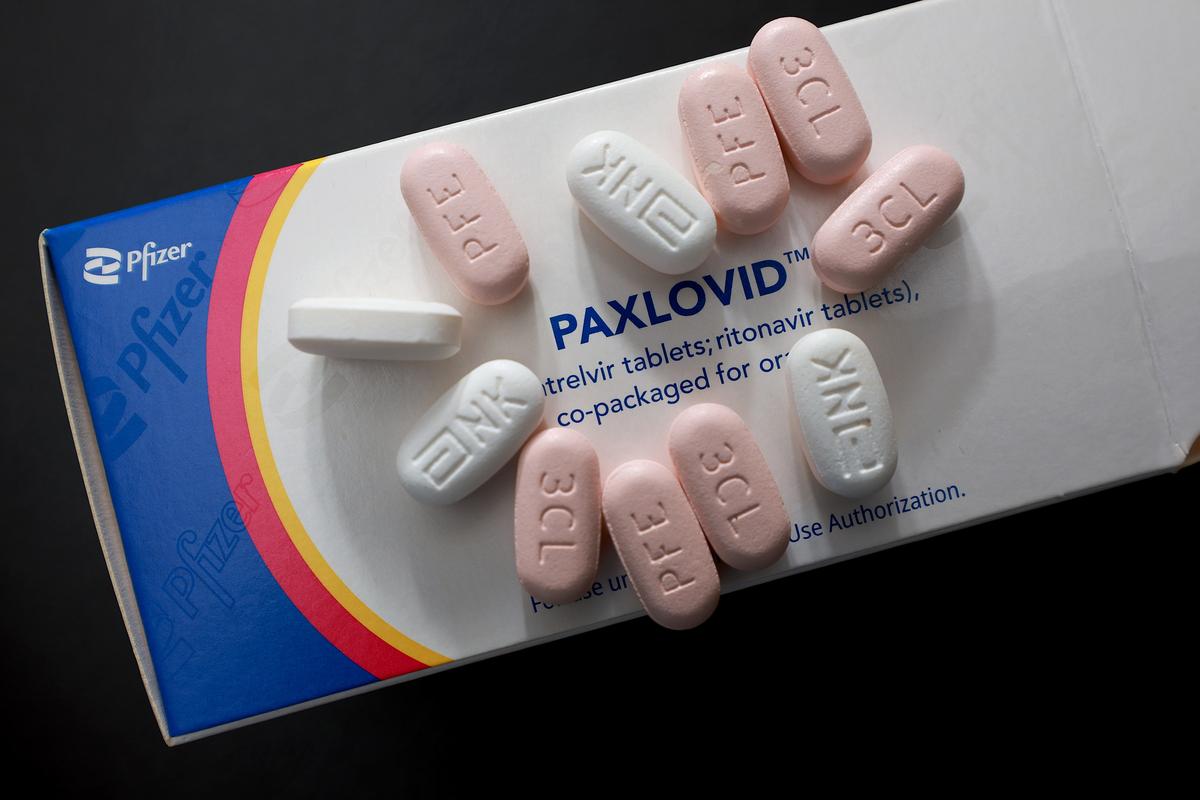
The U.S. Food and Drug Administration (FDA) on May 25 fully approved Pfizer’s oral antiviral medication Paxlovid to treat mild to moderate COVID-19 in adults at risk of severe infections.
This clears the way for the drugmaker to sell it at market rates once the U.S. government supplies dwindle.