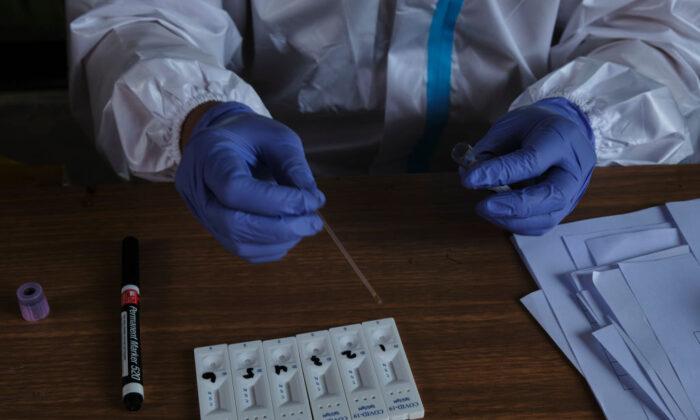
Australians will be able to get access to COVID-19 home rapid antigen test kits from November pending approval by regulator the Therapeutic Goods Administration (TGA) due in the coming month.
“This is an important additional protection for Australians: home testing to support Australians and to support the national plan,” Hunt said.
Rapid antigen at-home testing kits can return results in around 20 minutes and have been in use overseas for months.
TGA Deputy Secretary John Skerritt said he was confident that the home tests would be available for Australians in November.
However, he told a Senate committee that the TGA would be looking to make sure all home testing kits must be able to detect the Delta variant of the CCP virus.
“It does seem that some of the tests—and I don’t want to name particular products, but some of them are quite significant products—are much less sensitive against Delta,” he said.
Each of the tests will require TGA approval and inclusion in the Australian Register of Therapeutic Goods (ARTG).
“This is an important step in supporting the National Plan to transition Australia’s National COVID-19 Response and aligns with the timeframe where it is expected that approximately 70 percent of Australians will be double vaccinated,” the TGA said.
The TGA has noted that any tests approved will also need appropriate support available for consumers who are seeking advice or help with forms, including online videos, a 1800 call number, or a website fact sheet for consumers who are seeking help.
The TGA has also noted that prior to the release of the tests, state and territories will need to have appropriate systems in place to ensure any consumer who has a positive rapid antigen test result is supported to immediately have a confirmatory PCR test at a COVID-19 testing centre.
This could mean state and territory governments may need to implement strategies to compel people who test positive at home to get a PCR test.
Australian authorities had been cautious in approving the use of the testing kits for at-home use because of concerns around accuracy compared to nose and throat swabs.
However, Hunt is confident the kits will provide Australians with an additional layer of protection from the pandemic.
“One of the important things is that we can supplement what is known as PCR testing—the testing that we all know if we go to a Commonwealth or a state clinic—with the home testing,” he said.
Hunt noted that once approved, the rapid antigen tests would be available for purchase at pharmacies around Australia.