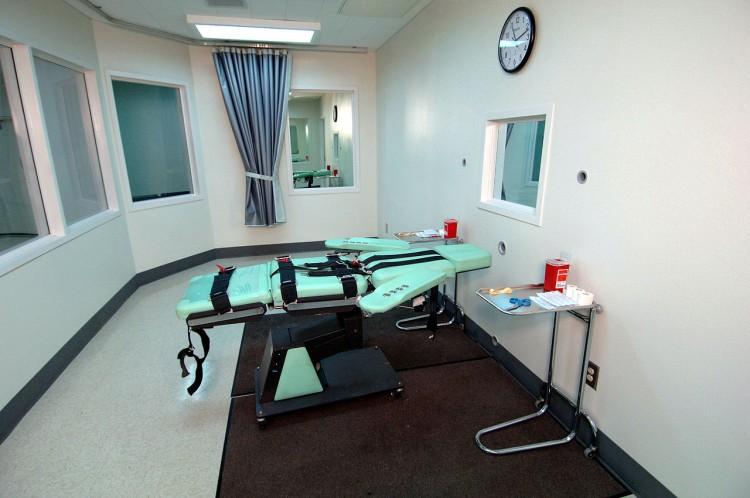
Lethal injection is the leading method of execution in the United States, but a sedative found in some capital punishment cocktails can only be found overseas. This week a federal court ruled to cut off the imported supply.
Sodium thiopental (better known by the trade name sodium pentothal) is a short-acting barbiturate used to induce anesthesia. Several states include thiopental in their lethal injection protocol, serving as an agent to render death penalty prisoners unconscious before other drugs take effect.
According to U.S. District Judge Richard Leon, because an imported drug may not function as intended, correctional facilities run the risk of subjecting prisoners to conscious pain, suffering, and cardiac arrest when using foreign thiopental.
In a ruling this week, Judge Leon ordered the U.S. Food and Drug Administration to round up state supplies of the imported sedative, and confiscate any shipments coming into the country.
The issue of foreign thiopental began in 2009, when Hospira Inc., the last U.S. pharmaceutical company to manufacture the drug, stopped production. Hospira had plans to move production to Italy until officials there realized what the drug might be used for.
While thiopental still sees some application outside of capital punishment, use among doctors has decreased with the introduction of newer medications, such as propofol—a drug, which several state correctional facilities, such as Ohio, Oklahoma, and Texas, now use in place of thiopental.
But when Hospira ended its production, other states weren’t willing to switch. Interested U.S. thiopental buyers found a market through Dream Pharma—a London wholesaler who bought the drug from an Austrian manufacturer.