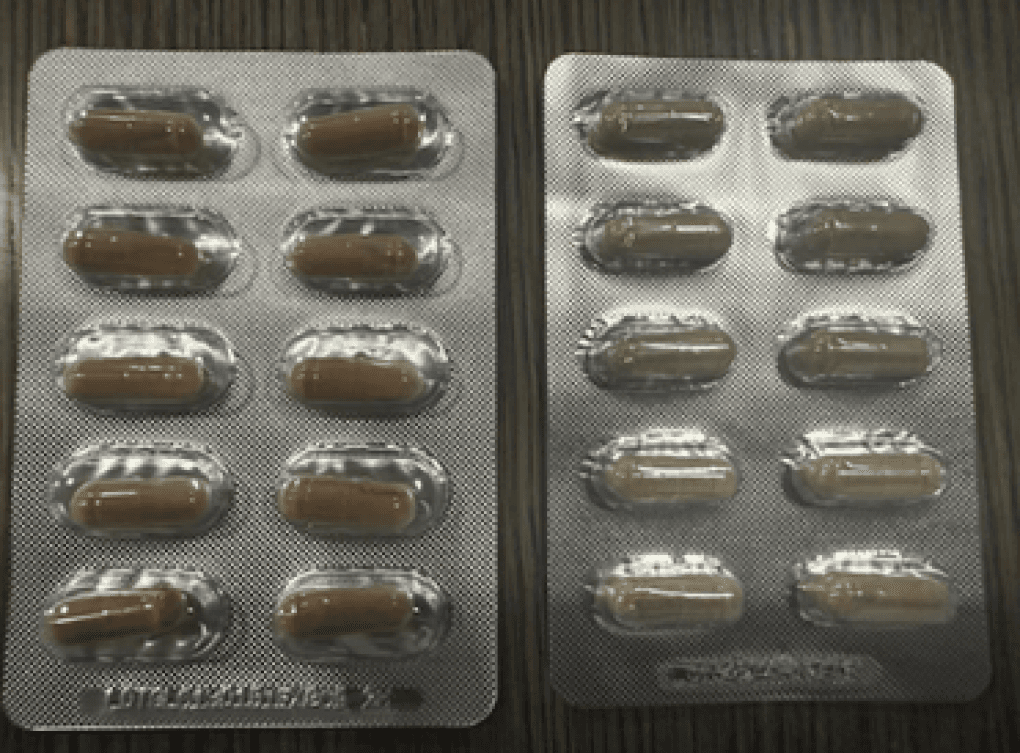
California-based Green Lumber Holdings is alerting consumers to counterfeit products misrepresented as genuine Green Lumber, an energy enhancement supplement, according to an Aug. 29 alert issued by the Food and Drug Administration (FDA).
The FDA found tadalafil, a prescription drug for erectile dysfunction, in products labeled as Green Lumber. Based on FDA findings, the company conducted an internal investigation and found that a company employee diverted legitimate packaging and customer information to distribute counterfeit, adulterated products.