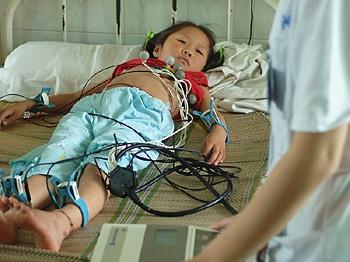
A 20-month-old infant in Fengtai died two days after receiving the company’s vaccine against Hepatitis A. As a result, the Health Services Bureau of Fengtai in Beijing suspended use of the vaccine.
Another girl from Chaoyang District resident died after a two-day fever, immediately following her injection with the same type of vaccine, the report stated, quoting the local newspaper Jinghua Times.
The suspended vaccine came from lot 2008052105, of which 19,850 doses have been distributed. The lot passed routine government safety inspections in July 2008, according to a statement released by Sinovac on December 2.
Authorities of Beijing Health Bureau said that the vaccines were bought through open bidding through proper channels and should have no quality problem. Therefore, there is no reason to stop the inoculation in the city, they argued.
Authorities from the Health Services Bureau of Chaoyang District maintained that the high fever of another girl was only an individual case and it cannot prove the vaccine has a quality problem.
The Fengtai baby’s body has been sent to an autopsy but the result of the autopsy is not yet known.
Read this article in the original Chinese: http://epochtimes.com/gb/8/12/7/n2354866.htm