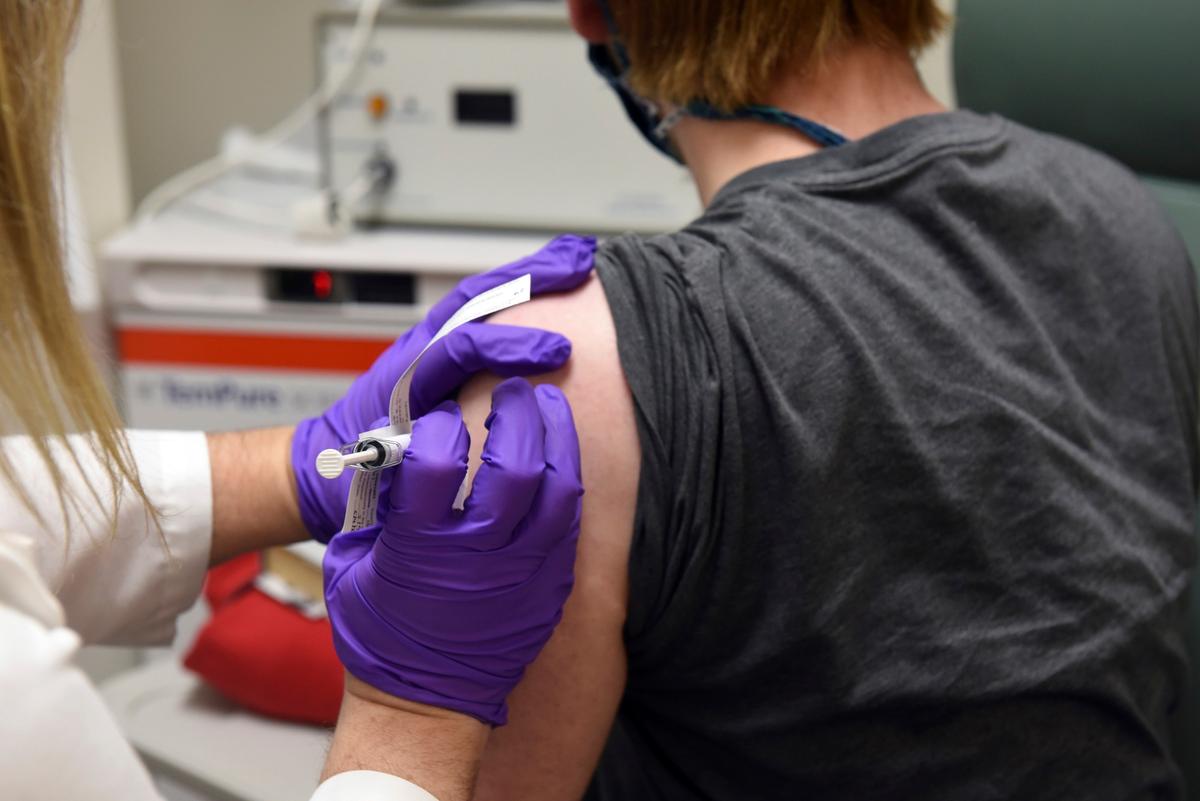
Pfizer and German partner BioNTech say their COVID-19 vaccine candidate is proving to be strongly effective in a large late-stage study, according to early data released on Nov. 9.
The phase three results, analyzed by an independent data monitoring board, indicate an efficacy rate above 90 percent at seven days after the second dose, the companies said. That means protection is achieved 28 days after the first vaccine. The vaccination schedule is two doses.