It is not clear when the FDA found what has been characterized as a “trace level” of melamine in the infant formula. It is also not clear why the information was released at this point.
Earlier this month, during a press conference announcing the opening of three FDA inspection offices in China, FDA officials were caught by surprise when their Chinese counterparts said they would also send their own inspectors to the U.S.
China’s stated intention to establish inspections on U.S. soil could be added pressure to domestic FDA operations.
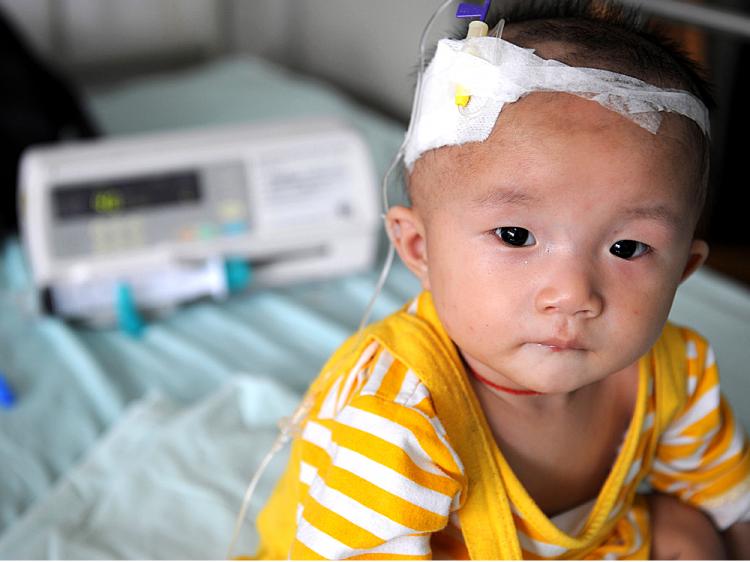
On Nov. 12, the FDA halted the import of all milk and milk-derived products from China. The move came nearly two months after a scandal over melamine-contaminated infant formula broke in China. More than 53,000 Chinese babies have been sickened by infant formula poisoned with the chemical. Several have died.
According to the FDA, melamine and melamine-analogs have been detected in a large number of products coming from China. Cyanuric acid, which forms a toxic compound when coupled with melamine, has also been detected in various products. Affected products will be detained at border entry points until they have been tested by third-party laboratories.
The FDA has said that the extraordinary step was taken “to protect the health of the U.S. consumers.”
“Our testing has found melamine or melamine analogs in a variety of food products manufactured in China that contain milk or milk-derived components,” said Mike Herndon, an FDA Press Officer.
More than 50 milk or milk-derived products, milk-derived ingredients, and finished food products containing milk are now flagged by the FDA for possible melamine and cyanuric acid contamination. The list of products includes candy, beverages, bakery products, and baby food.
All milk products, all milk-derived ingredients, and finished food products containing milk from China will be detained until they have been tested by third-party laboratories. Importers must show through the testing that at least 5 consecutive shipments meet the minimum standard for melamine and cyanuric acid. The FDA allows 2.5 parts per million (ppm) of melamine in food for adult consumption.
The FDA did not release any brand names associated with the contaminated baby formula found in the U.S. However, the aim in releasing the information might have been part of a larger plan to establish a minimum level of melamine considered to be acceptable, despite the lack of scientific knowledge about melamine’s impact on infants.
“I hope they don’t use this as an excuse to set a standard for infants,” says Tony Corbo, a lobbyist with health and consumer advocacy group Food and Water Watch in Washington, D.C.
Corbo adds that it won’t be that simple for the FDA to establish a risk standard for melamine in infant formula, since their original position was one of “zero tolerance”.
In an Oct. 3 safety assessment, the FDA stated it couldn’t establish a “safe level of melamine and melamine-related compounds in infant formula that does not raise public health concerns”. A gap in scientific knowledge about melamine’s toxicity and analogues in infants were cited as reasons for this inability.
FDA Interim Safety Risk Assessment on Melamine
http://www.fda.gov/bbs/topics/NEWS/2008/NEW01895.html