An unusual family of minerals could be used in the computer chips of the future to hold more data and encrypt it more securely, says a team of scientists collaborating in the U.S. and U.K.
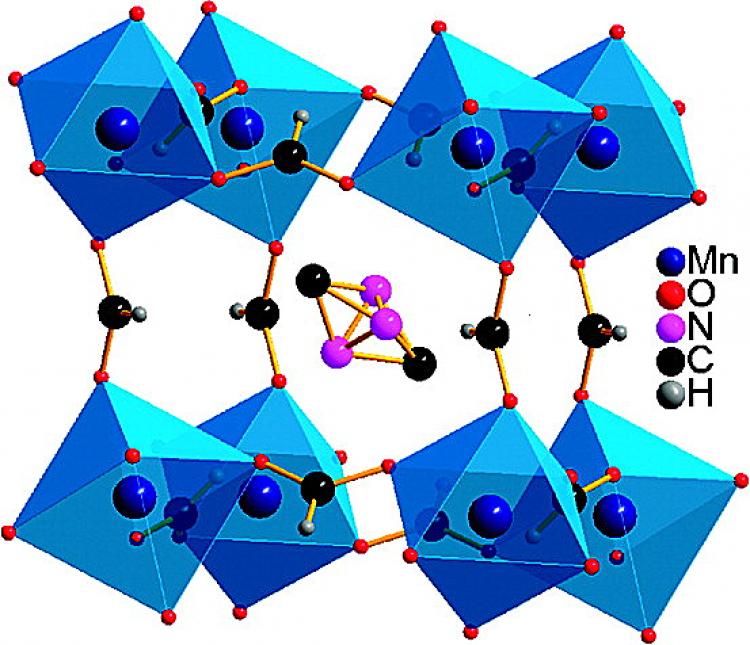
Using laboratory experiments and computer analysis, the researchers found that four of the porous crystalline minerals known as “metal-organic frameworks” (MOFs) possessed rare dual properties when cooled to a specific temperature—that is, they were “multiferroic.”
This means that the crystals have magnetic poles (ferromagnetic) and at the same time a positive and negative electrical charge that can be reversed when an external electrical field is applied (ferroelectric).
“Normally, these two properties are mutually exclusive,” said Dr. Naresh Dalal, professor at Florida State University and researcher at the National High Magnetic Field Laboratory, in a press release.
The researchers found that the multiferroic properties were the result of hydrogen bonds forming between the components of the MOFs—a new concept in the field of multiferroics.
“Finding four multiferroic materials at one time is quite scientifically significant and opens numerous doors in terms of potential applications.”
These applications could include the boosting of the data storage capacity of computer memory and other data storage devices, according to the researchers.
“Theoretically, it might be possible to design devices that are much smaller and faster than the ones we use today to store and transmit data,” said Dr. Harold Kroto, professor at Florida State University. “And with data split over two mediums, information could be encrypted in a way that makes it far more secure than is currently possible. This could have wide-ranging applications in areas as diverse as the aeronautics industry, the military, the workplace, and even the average consumer’s home.”
Another advantage of using the crystals in high-tech devices is the environmental and health benefits, said Dalal.
“The four new multiferroic crystals that we have identified all substitute other, less toxic metals for lead, which is a potent neurotoxin,” he said. “By reducing the amount of lead that enters landfills, we also reduce the amount that enters our water supply—and our bodies.”
But before any of this can happen, scientists would have to figure out how to significantly raise the temperature at which the multiferroic properties emerge to around room temperature, commented Dr. Ramamoorthy Ramesh of the University of California, Berkeley, in the journal Nature.
The research is published in the Journal of the American Chemical Society. It is an extension of three decades of work by Dr. Dalal and was instigated by Dr. Kroto, who received the 1996 Nobel Prize for Chemistry and Biochemistry for co-discovering pure carbon “footballs,” or buckminsterfullerene.
To read the research paper, please visit http://pubs.acs.org/doi/full/10.1021/ja904156s
Using laboratory experiments and computer analysis, the researchers found that four of the porous crystalline minerals known as “metal-organic frameworks” (MOFs) possessed rare dual properties when cooled to a specific temperature—that is, they were “multiferroic.”
This means that the crystals have magnetic poles (ferromagnetic) and at the same time a positive and negative electrical charge that can be reversed when an external electrical field is applied (ferroelectric).
“Normally, these two properties are mutually exclusive,” said Dr. Naresh Dalal, professor at Florida State University and researcher at the National High Magnetic Field Laboratory, in a press release.
The researchers found that the multiferroic properties were the result of hydrogen bonds forming between the components of the MOFs—a new concept in the field of multiferroics.
“Finding four multiferroic materials at one time is quite scientifically significant and opens numerous doors in terms of potential applications.”
These applications could include the boosting of the data storage capacity of computer memory and other data storage devices, according to the researchers.
“Theoretically, it might be possible to design devices that are much smaller and faster than the ones we use today to store and transmit data,” said Dr. Harold Kroto, professor at Florida State University. “And with data split over two mediums, information could be encrypted in a way that makes it far more secure than is currently possible. This could have wide-ranging applications in areas as diverse as the aeronautics industry, the military, the workplace, and even the average consumer’s home.”
Another advantage of using the crystals in high-tech devices is the environmental and health benefits, said Dalal.
“The four new multiferroic crystals that we have identified all substitute other, less toxic metals for lead, which is a potent neurotoxin,” he said. “By reducing the amount of lead that enters landfills, we also reduce the amount that enters our water supply—and our bodies.”
But before any of this can happen, scientists would have to figure out how to significantly raise the temperature at which the multiferroic properties emerge to around room temperature, commented Dr. Ramamoorthy Ramesh of the University of California, Berkeley, in the journal Nature.
The research is published in the Journal of the American Chemical Society. It is an extension of three decades of work by Dr. Dalal and was instigated by Dr. Kroto, who received the 1996 Nobel Prize for Chemistry and Biochemistry for co-discovering pure carbon “footballs,” or buckminsterfullerene.
To read the research paper, please visit http://pubs.acs.org/doi/full/10.1021/ja904156s