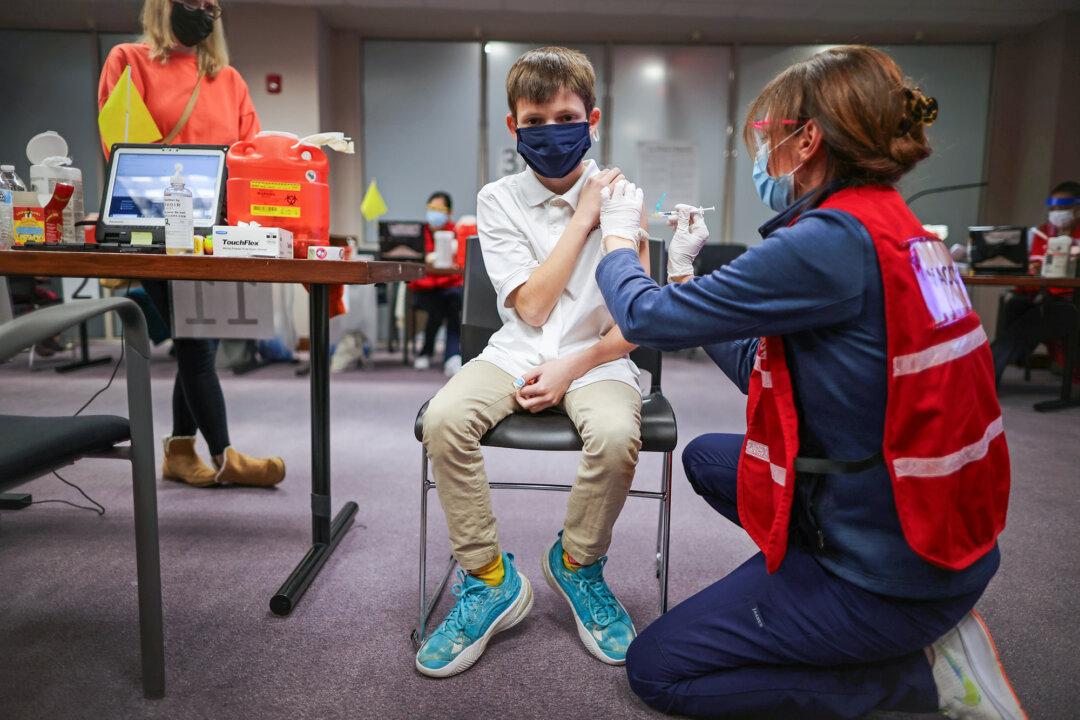
President Joe Biden’s administration is planning a large-scale rollout of COVID-19 vaccines for children as young as 5 years old, officials announced on Oct. 20.
The administration has secured enough doses of the Pfizer-BioNTech vaccine to vaccinate all of the country’s 28 million children between the ages of 5 and 11 and is prepared to ship the doses across the country to facilitate vaccination efforts, the White House stated.