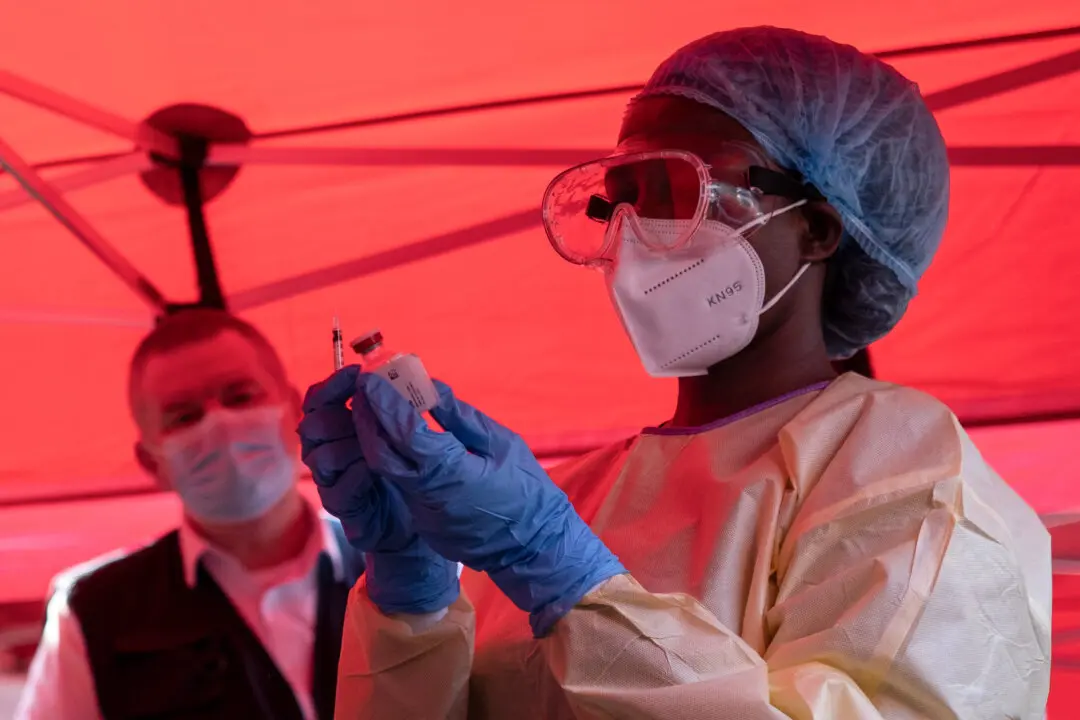
U.S. authorities are preparing to ship out millions of doses of Pfizer’s COVID-19 vaccine by the end of February if drug regulators authorize the shot for children under 5.
President Joe Biden’s administration has secured enough doses to vaccinate all 18 million children in the country who are 4 or younger. Officials are telling health care providers to pre-order shots.