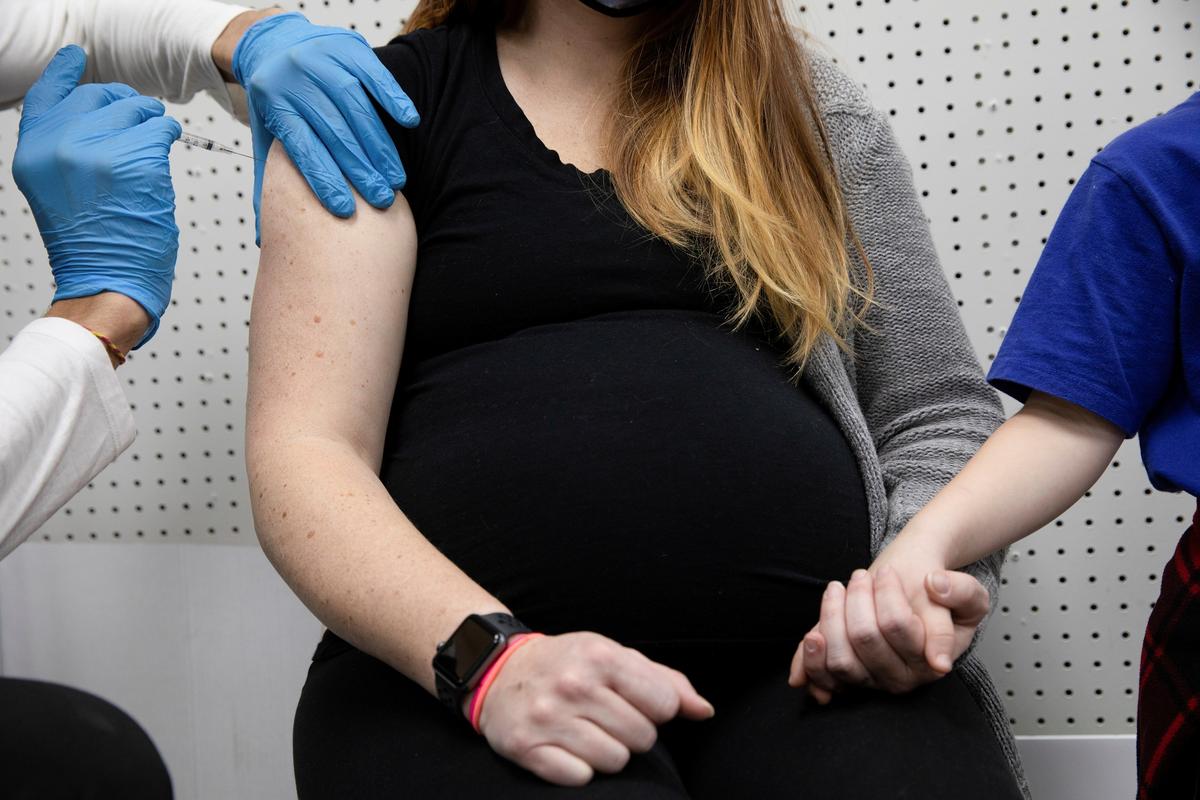
The Centers for Disease Control and Prevention (CDC) is recommending that pregnant women receive a COVID-19 vaccination.
“CDC recommends that pregnant people receive the COVID-19 vaccine. We know that this is a deeply personal decision, and I encourage people to talk to their doctors or primary care providers to determine what is best for them and for their baby,” Dr. Rochelle Walensky, the health agency’s head, said in a virtual briefing on April 23.