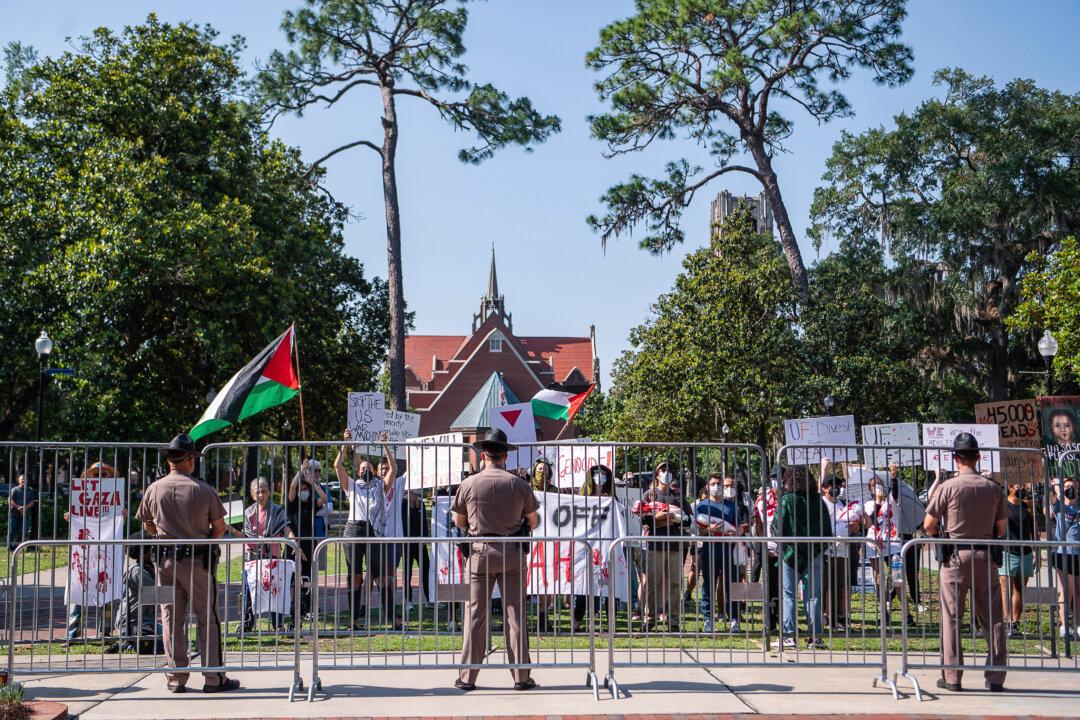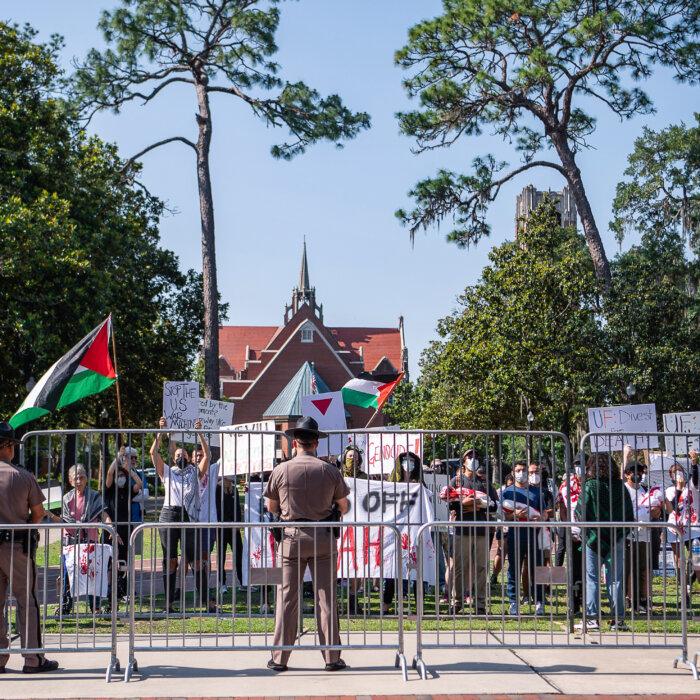SPECIAL COVERAGE
Read More
Read More
Georgia Appeals Court Agrees to Review Fani Willis Disqualification Ruling
Should Ms. Willis and her office be removed from the case, a state board would need to appoint a new prosecutor.
Georgia Appeals Court Agrees to Review Fani Willis Disqualification Ruling
Should Ms. Willis and her office be removed from the case, a state board would need to appoint a new prosecutor.
Trending Videos
Top Premium Reads
Top Stories
Most Read
House Approves 1-Week Extension for FAA Reauthorization
The vote comes after many expected the Senate to miss its original May 10 deadline.
House Panel Subpoenas Top Fauci Adviser to Appear and Answer COVID Questions
U.S. National Institutes of Health official has been stonewalling lawmakers, panel chairman says.
US Delaying Some Weapons Shipments to Israel, Austin Confirms
‘We are currently reviewing some near-term security assistance shipments in the context of the unfolding events in Rafah,’ Defense Secretary Lloyd Austin said.
Activists Highlight Persecution in China During CCP Leader’s Trip to Europe
Rights groups called on the EU to protect its citizens and institutions from ‘complicity in aiding and abetting forced organ harvesting’ in communist China.
DeSantis Touts Florida Response to Pro-Palestianian Campus Protests
The governor praised local authorities for maintaining ‘law and order’ amid escalating campus protests across the country.
Police Clear GWU Pro-Palestinian Encampment, Prompting Cancellation of Congressional Hearing
The operation occurred at 3:45 a.m. EST, just hours before Washington Mayor Bowser and Police Chief Smith were scheduled to testify before Congress.
Chris Cuomo Backtracks on Vaccine Injury Suggestion, Touts Ivermectin
Former CNN host said he was ’sick' like people injured by COVID-19 vaccines.
Biden Campaign Intensifies Presence in Battleground States
‘Trump’s paid media strategy can only be described as anemic and inefficient,’ Biden campaign communications director Michael Tyler told the press.
Oakland Restaurants Boost Security, but Thieves Won’t Back Off
‘They’ll break into cars in broad daylight with the police in the lot,’ says one employee.
Editors' Picks
US Government, Lawmakers Condemn Russia’s Detention of Falun Gong Practitioner
US officials said they are ‘troubled and saddened’ to hear of the Moscow police raid and ‘deeply concerned’ about the persecution of Falun Gong globally.
EXCLUSIVE: Inside the Study that Shook the CDC
Study that detailed sudden deaths in two teenagers following COVID-19 vaccination left officials scrambling.
Biden Admin to Exclude Avian Influenza, COVID-19 From Risky Research Rules
Avian influenza strain H5N1 and the Ebola virus are not considered to be potential pandemic pathogens by the National Science and Technology Council.
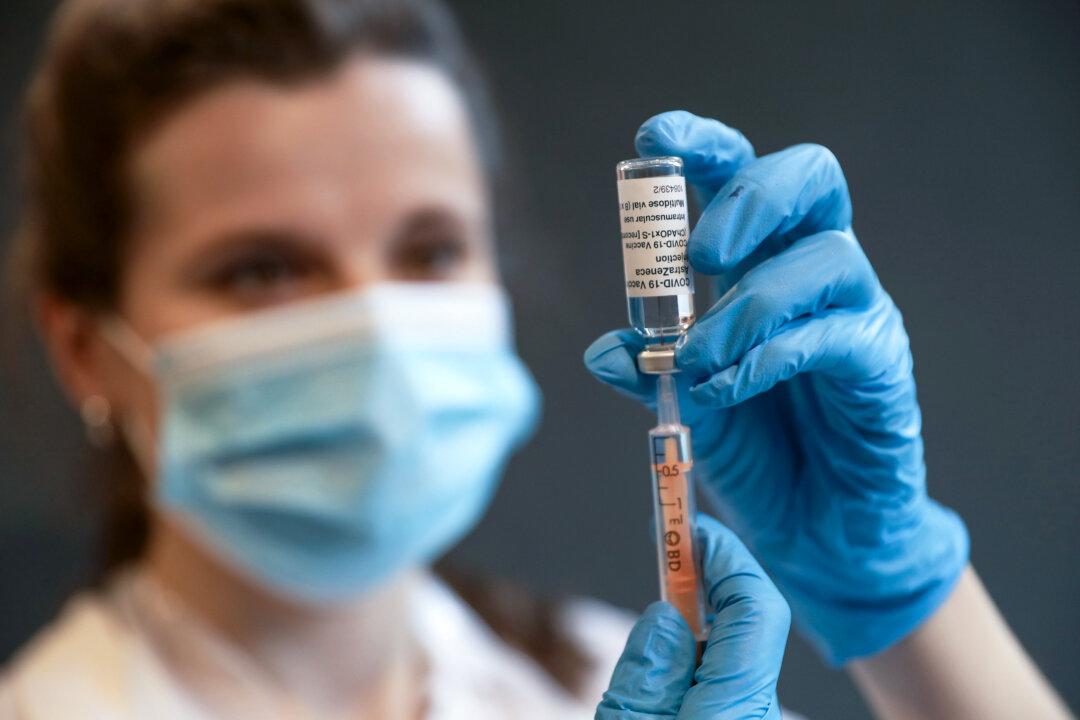
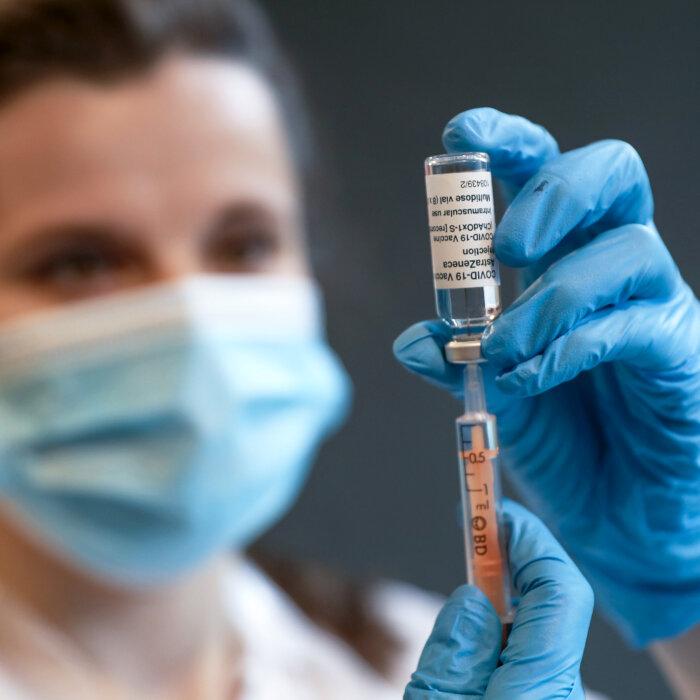
AstraZeneca Begins Worldwide Withdrawal of COVID-19 Vaccine
AstraZeneca recently admitted in a UK court document that its shot ‘can, in very rare cases, cause’ blood clots and low platelets.

Why We Need Poetry: Seeing the World Anew
It’s the poet’s vocation to see the world’s beauty and mystery.
Why We Need Poetry: Seeing the World Anew
It’s the poet’s vocation to see the world’s beauty and mystery.
Epoch Readers’ Stories
A History Of The American Nation
A patriotic poem by Ted Schneider
Of Cars and Kids
Why should our kids have to settle for a Trabant, or a Pyonghwa, education when they could have a BMW?
A Nation Divided
Poem by an American Patriot
What Is Going on Here?
There are two major things plants need to survive and continue generating our life saving oxygen. The first is CO2, and the second is sunshine.
Inspired Stories
Empower the World with Your Story: Share Love, Inspiration, and Hope with Millions
Special Coverage
Special Coverage

5 Exercises to Alleviate Digestive Discomfort
Indigestion? Try these five exercises to soothe the intestines and stomach, alleviating discomfort caused by overeating.

5 Exercises to Alleviate Digestive Discomfort
Indigestion? Try these five exercises to soothe the intestines and stomach, alleviating discomfort caused by overeating.

Edith Meiser: America’s Greatest Sherlockian
In this installment of ‘Profiles in History,’ we meet the writer and actress who popularized Sherlock Holmes in America via a different media.

The Newberry Opera House: Music and More for 142 Years
In this installment of ‘History Off the Beaten Path,’ we visit a tiny, rural South Carolina opera house, where performers still take the stage.

Hollywood Legend Gregory Peck: A Dignified Man On-Screen and Off-Screen
Gregory Peck’s moral convictions defined the type of roles he wanted to play on screen.

Hans Sachs: Where Shoemaking Meets Songwriting
The Meistersinger guild of blacksmiths, carpenters, lawyers, teachers, and businessmen wrote and recited songs. Hans Sachs was the greatest of them.

‘Legends of Liberty 2’: A Fabulous Sequel
Andrew Benson Brown has reached new heights of poetic achievement with his second volume of ‘Legends of Liberty.’

Pythagoras To Beethoven: A Hillsdale College Free Online Music Course
“Join me at the piano,” says concert pianist Hyperion Knight for a free, online course on classical music through Hillsdale College.

Brian Kilmeade’s Love of America and Defense of Its History
The ‘Fox & Friends’ host has written six books that highlight American heroes who put their love of country before their own interests.

Edith Meiser: America’s Greatest Sherlockian
In this installment of ‘Profiles in History,’ we meet the writer and actress who popularized Sherlock Holmes in America via a different media.

This Springtime Dessert Actually Tastes Like Strawberries
A strawberry-sour cream topping gives these cheesecake bars extra flavor without sacrificing creaminess.

Ed Perkins on Travel: Amtrak’s Surprise
A promising new long-haul train from Amtrak assures on time arrivals on trips between Chicago and the Twin Cities.

Rick Steves’ Europe: Madrid’s Outdoor Delights
Madrid is best enjoyed on the car-less streets and after the sun sets, when the temperature is more bearable.

Why Adult Children Are Finding Benefits to Traveling With Their Parents
Adult children are giving back to their parents with trips and adventures.
Ed Perkins on Travel: Amtrak’s Surprise
A promising new long-haul train from Amtrak assures on time arrivals on trips between Chicago and the Twin Cities.
























































![[PREMIERING MAY 9, 8:30PM ET] Weapons of Mass Migration | NEW Documentary](/_next/image?url=https%3A%2F%2Fimg.theepochtimes.com%2Fassets%2Fuploads%2F2024%2F05%2F04%2Fid5643663-900x1350-600x900.jpg&w=1200&q=75)