Health authorities in Europe, North America, and Australia have begun to investigate an outbreak of monkeypox, raising concerns that a broader outbreak of the viral infection could be underway in the West.
The CDC has issued a recommendation of “intensive health measures” for the UK, including contact tracing, case searching, and rash-illness surveillance, as well as the isolation of individuals suspected of being infected with the disease.
The monkeypox virus was first reported among laboratory monkeys in 1958, with the first human cases recorded in 1970 in the Democratic Republic of the Congo. Typically, the disease has been confined to its African homeland, and while cases have sporadically been reported among individuals traveling from Africa to the United States or Europe in recent years, the prevalence of monkeypox in the West is unprecedented, suggesting the virus has taken hold in Europe.
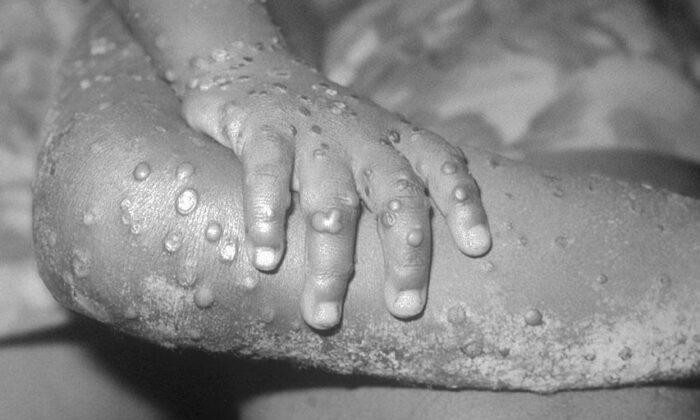
Symptoms include fever, body aches, and rashes. Though related to the smallpox virus, symptoms are typically less severe for monkeypox. The latter is notably distinguished from smallpox by the appearance of swollen lymph nodes during the symptomatic phase of the virus, immediately preceding a swollen rash that spreads to the inside of the mouth and the hands and feet.
Transmission occurs primarily through large airborne saliva droplets, and therefore requires prolonged exposure to an infected person in order to spread.
Vaccines for the monkeypox virus already exist, and the U.S. Biomedical Advanced Research and Development Authority, part of the U.S. Department of Health and Human Services, moved to secure doses on May 18 after discovering its first case in the United States in Massachusetts.
Danish biotechnology company Bavarian Nordic, maker of the Jynneos smallpox vaccine, said in a statement that the United States “exercised the first options under the contract to supply a freeze-dried version” of the vaccine, allowing for the first doses to be available in 2023 and 2024. Freeze-drying provides a longer shelf life.
Paul Chaplin, president and CEO of Bavarian Nordic said: “This marks a significant milestone in our long-standing partnership with the U.S. government to ensure availability of life-saving vaccines for the entire population.”






Friends Read Free