SPECIAL COVERAGE
Read More
Read More
Takeaways From Week 1 of Trump New York Trial
When trial begins next week, attorneys for former President Donald Trump won’t even know what witnesses prosecutors will call to the stand until they enter the
Takeaways From Week 1 of Trump New York Trial
When trial begins next week, attorneys for former President Donald Trump won’t even know what witnesses prosecutors will call to the stand until they enter the
Trending Videos
Top Premium Reads
Top Stories
Most Read
Trump Attorneys Fight to Prevent Past Cases From Being Brought Up in Trial
Trump confirmed he was willing to testify, despite the prosecutor’s push to bring up past cases.
Comer Threatens to Subpoena Biden Energy Secretary After She Finds Time for ‘The View’ But Not Congress
The House Oversight Committee chairman has been pressing the Energy secretary for details on the decision to halt new liquid natural gas (LNG) exports.
IRS Announces Tax Refund Increase as It Rakes in Near-Record $4.7 Trillion From Taxpayers
Tax refunds boosted by 3 percent.
Prosecutor Says Charges Dropped Against Election Software Company for Political Reasons
A tort claim alleges Los Angeles DA George Gascon dropped the case after being praised by former President Donald Trump.
Senate Scrambles to Reach FISA Deal Before Midnight Deadline
Members are divided over warrantless spying powers that have been used to target U.S. citizens.
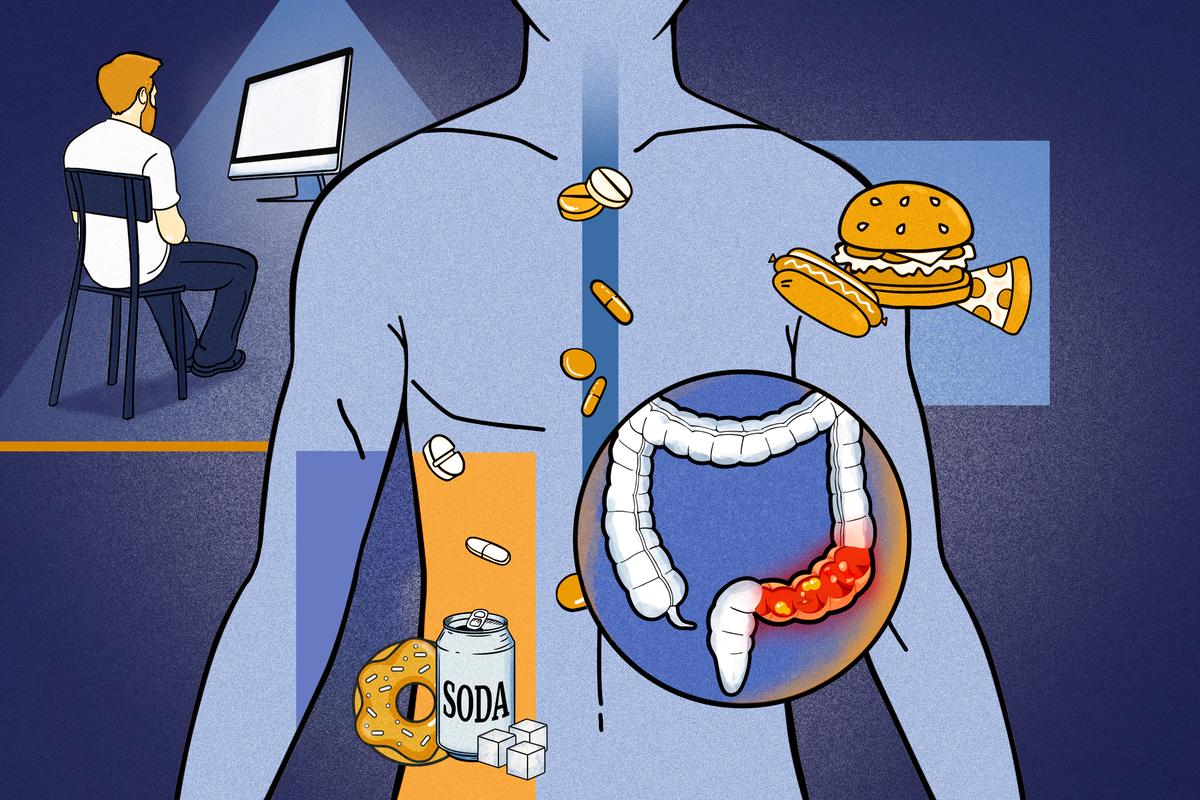
Young Kids Nearly 5 Times More Likely to Be Hospitalized With RSV After COVID Pandemic
A new study links the COVID-19 pandemic to a seasonal shift and more severe resurgences of respiratory syncytial virus in children younger than 5.
Planned Parenthood Abortions Among ‘Top Four Leading Causes of Death’ in America
The organization reduced the number of consultations on health issues, while it focused on providing abortion services.
IRS Sued for Illegally ‘Concealing’ Records on ‘Race-Based Tax Audits’
America First Legal filed the complaint earlier this week.
Man Sets Himself on Fire Outside Trump Trial Courthouse
The act of self-immolation lasted about 45 seconds and ended when police put the flames out with a fire extinguisher.
Biden Admin’s Anti-Sex Discrimination Rule Redefines Sex to Include Gender Identity
The new rule overturns the Trump-era rule on how schools respond to complaints of sexual misconduct.
Top Military Official Lied About Jan. 6: Whistleblowers
Army Secretary Ryan McCarthy was among senior officials who did not tell the truth about what happened on Jan. 6, 2021, National Guard officials say.
Biden Restricts New Oil and Gas Leasing on 13 Million Acres of Alaskan Land
‘When you take off access to our resources ... this is the energy insecurity that we’re talking about,’ Rep. Lisa Murkowski (R-Alaska) said.
NatCon Cancellation Attempt Shows Rising Threat to Free Speech in Europe
‘Don’t vacate the premises. Make them throw you out,’ ADF International’s Jean-Paul Van De Walle said. ‘Make them show what they’re capable of doing.’
Ukraine, Israel Aid Bill Clears Procedural Hurdle in House With Democrat Help
In a move breaking from custom, Democrats stepped in to help advance aid bills for Ukraine, Israel, and Taiwan, and a measure that could ban TikTok.

Celebrating the World’s Best Portrait and Figurative Artworks
The Portrait Society of America selected its International Portrait Competition finalists.
Celebrating the World’s Best Portrait and Figurative Artworks
The Portrait Society of America selected its International Portrait Competition finalists.
Epoch Readers’ Stories
Wisdom From a Retired Cowboy Artist
I have lived the life of the sculptures I have made.
Section 3 of 14th Amendment Not Applicable for Ruling Trump out of 2024
I don’t believe that Section 3 of 14th Amendment is self-executive and therefore can not be applied directly by the court.
Inspired Stories
Empower the World with Your Story: Share Love, Inspiration, and Hope with Millions
Special Coverage
Special Coverage

Parkinson’s Dulls Physical Response to Emotions: Study
A new study has suggested that anger, disgust, and even sadness hit with less force for those who have the neurodegenerative disorder.

Parkinson’s Dulls Physical Response to Emotions: Study
A new study has suggested that anger, disgust, and even sadness hit with less force for those who have the neurodegenerative disorder.

The Surprising Roots of Indoctrination by Colleges
A West Virginia University professor names names and pinpoints how college indoctrination began, in ‘Winning America’s Second Civil War.’

‘God Bless the U.S.A.’
Lee Greenwood’s surprise single release became a classic American anthem and a unifying symbol of patriotism.

The American Mozart: Composer Timothy Olmsted
Timothy Olmsted’s lifelong dedication to music made him one of the country’s earliest influential musicians.

S. Truett Cathy: Founder of Chick-fil-A
This entrepreneur called on his ethic of hard work to build a successful fast food franchise.

Rembrandt’s Sole Seascape and the Great Heist
Stolen from the Isabella Stewart Gardner Museum in 1990, ‘Christ in the Storm on the Sea of Galilee’ along with two other Rembrandts are still missing.

Chicken Shawarma in a Bowl Is a Tasty, Healthy Meal
Roasting the chicken legs while the grain is cooking will help this dish come together quicker.

Taking the Kids: And Heading to Less Visited National Parks
There are 429 national park sites in the U.S., though just 63 have the “National Park” designation in their names.

Thailand Leads Push for Six-Nation Visa to Lure Moneyed Tourists
The plan is so that tourists can travel six countries on one visa, but some officials are not optimistic.

Brazil Postpones Visa Requirements Once Again for American Visitors
The goal is to ensure that Brazil’s electronic visa program is fully up and running before applying the visa requirement.
Taking the Kids: And Heading to Less Visited National Parks
There are 429 national park sites in the U.S., though just 63 have the “National Park” designation in their names.




















































































































































![[LIVE 4/26 at 10:30AM ET] New Push Started for Global Digital Currencies](/_next/image?url=https%3A%2F%2Fimg.theepochtimes.com%2Fassets%2Fuploads%2F2024%2F04%2F19%2Fid5633115-0426-600x338.jpg&w=1200&q=75)